
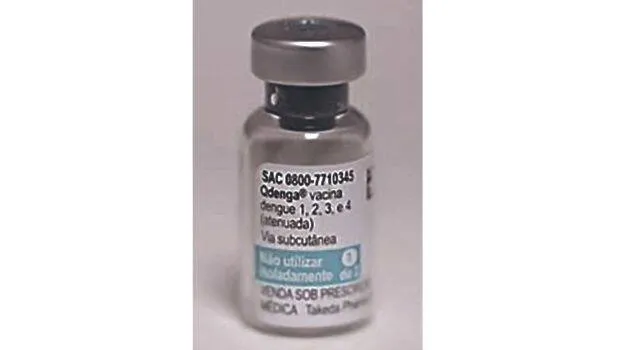
NEW DELHI: India will import the dengue vaccine 'Qdenga' (TAK-003) from Japan. The expert committee of the Central Drugs Standard Control Organisation (CDSCO) has approved the proposal. However, the vaccine will be given to people aged 4 to 60 only after final approval from the Drug Controller General of India. The vaccine has been developed by the Japanese biopharmaceutical company Takeda.
In India, clinical trials were conducted on 480 people aged between 4 and 60, and the vaccine was found to be safe. It is currently the only approved vaccine against dengue in the world and has an efficacy of around 64 percent. The vaccine has already been approved in 41 countries, including those in the European Union and the United Kingdom. It will be administered in two doses with a gap of three months. For children aged 6 to 16, the vaccine can be given without prior testing.