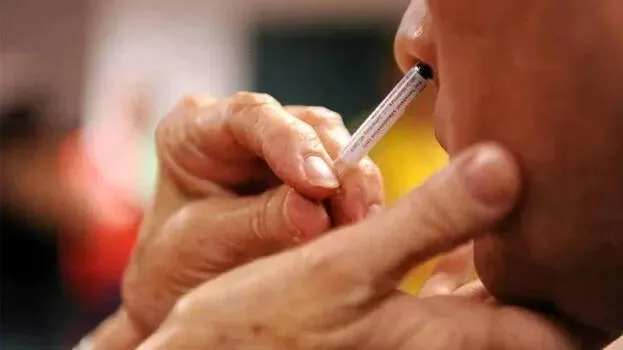
NEW DELHI: Bharat Biotech has successfully completed the third phase of the trial for Covid nasal vaccine. The company said it would submit the test results to the DCGI (Drug Controller General of India) next month.
‘The information is being analysed. The test results will be submitted to the DCGI shortly. If approved, it will be the world's first proven nasal Covid vaccine,’ said Dr Krishna Ella, Chairman and MD, Bharat Biotech.
The DCGI approved the third phase of testing for nasal vaccine in last January. The indications are that this vaccine is capable of resisting newer Covid variants such as Omicron.
Bharat Biotech is the second company in India to apply for the third phase trial of Covid vaccine. Krishna Ella said that those who have taken the two doses of vaccine must take the booster dose and the third dose will give better immunity.